Molnupiravir / Antivirale Therapie Positives Und Negatives Zu Molnupiravir Bei Covid 19 Pz Pharmazeutische Zeitung
Molnupiravir is the first oral direct-acting antiviral shown to be highly effective at reducing nasopharyngeal SARS-CoV-2 infectious virus and viral RNA and has a favorable safety and tolerability profile. Molnupiravir has been shown to be active in several preclinical models of SARS-CoV-2 including for prophylaxis treatment and prevention of transmission.
21 hours agoMolnupiravir is a nucleoside-analog a type of antiviral that has been effective against other viruses.

Molnupiravir. Molnupiravir was originally positioned as an anti-influenza drug. 1 day agoAmong patients taking molnupiravir 73 were either hospitalized or died at the end of 30 days compared with 141 of those getting the dummy pill. Molnupiravir increases the frequency of viral RNA mutations and impairs SARS-CoV-2 replication in animal models and in humans.
Of the participants who received molnupiravir 28 or 73. 9 hours agoMolnupiravir is a small molecule that wallops the work of a viral RNA-dependent RNA polymerase an enzyme critical for making copies of RNA viruses such as SARS-CoV-2. Molnupiravir MK-4482 is designed to induce viral genome copying errors to prevent the virus from replicating in the human body and evidence to date from clinical trials in patients with COVID-19 suggests that molnupiravir may reduce replication of the SAR-CoV-2 virus.
The pill developed by Merck and Ridgeback Biotherapeutics molnupiravir would be the first oral medication to treat the coronavirus if authorized by US. Molnupiravir EIDD-2801MK-4482 is an investigational orally bioavailable form of a potent ribonucleoside analog in development for the treatment of COVID-19. Molnupiravir MK-4482 EIDD-2801 is an experimental oral antiviral developed initially to treat influenza at Emory University.
So the mechanism here makes sense said Dr. Molnupiravir delivers a precise blow to viral RNA polymerase by posing as a building block for RNA. Molnupiravir has been shown to be active in several.
Oral molnupiravir MK-4482 EIDD-2801 is an experimental drug compound developed at Emory University USA and licensed by Ridgeback Biotherapeutics. 16 hours agoA drug like molnupiravir the name is a reference to Thors hammer Mjölnir could also help compensate for persistent gaps in Covid-19 vaccination coverage both in. Once that process is underway the drug inserts errors into the genetic code.
In a trial of 775 patients with mild-to-moderate COVID-19 who were considered higher risk for severe disease molnupiravir reduced hospitalization by. 20 hours agoMolnupiravir tricks the coronavirus into using the drug to try to replicate the viruss genetic material. Molnupiravir MK-4482 Antiviral Description.
1 day agoMolnupiravir MK-4482EIDD-2801 is an investigational orally administered form of a potent ribonucleoside analog that inhibits the replication of SARS-CoV-2 the causative agent of COVID-19. Molnupiravir was generally well tolerated with similar numbers of adverse events across all groups. Molnupiravir being a prodrug of the synthetic nucleoside analogue β-D-N 4 -hydroxycytidine.
Jha the dean of Brown. 22 hours agoMolnupiravir co-developed with Ridgeback Biotherapeutics is administered orally and works by inhibiting the replication of the coronavirus inside the body. An interim analysis of a Phase-3 study based on 775 trial.
There were no deaths in the drug group after. Molnupiravir works as an antiviral agent by inhibiting the replication of the SARS-CoV-2 virus the causative agent of COVID-19. Molnupiravir is an orally available antiviral drug candidate currently in phase III trials for the treatment of patients with COVID-19.
20 hours agoMolnupiravir is a small molecule that wallops the work of a viral RNA-dependent RNA polymerase an enzyme critical for making copies of RNA viruses such. Molnupiravir originally created by researchers at Emory University in Atlanta is given as four pills taken twice a day for five days. Molnupiravir is an orally bioavailable form of a potent ribonucleoside analog that inhibits replicating multiple RNA viruses including SARS-CoV-2 the causative agent of COVID-19.
Molnupiravir is being developed by Merck Co in collaboration with Ridgeback Biotherapeutics a US-based biotechnology company. 16 hours agoMolnupiravir is the first oral antiviral to have shown efficacy in the outpatient setting for COVID said Daria Hazuda Mercks vice president for infectious disease and vaccine discovery. 5 hours agomolnupiravir is also being evaluated for whether it can help prevent transmission of virus or as prophylaxis in MOVe-AHEAD a global multicentre randomised double-blind placebo-controlled.
2 Molecular echanism of molnupiravir-induced SARS-CoV-2 mutagenesis. Molnupiravir if approved would be the first orally active direct-acting antiviral drug for COVID a significant advance in fighting the pandemic. Molnupiravir is an experimental antiviral drug that is orally active and was originally developed for the treatment of influenza.
The companies plan to seek emergency authorisation for the drug in the US as soon as possible the company said in a statement.

Neue Studiendaten Molnupiravir Schutzt Mause Vor Corona Infektion Und Cov Pz Pharmazeutische Zeitung
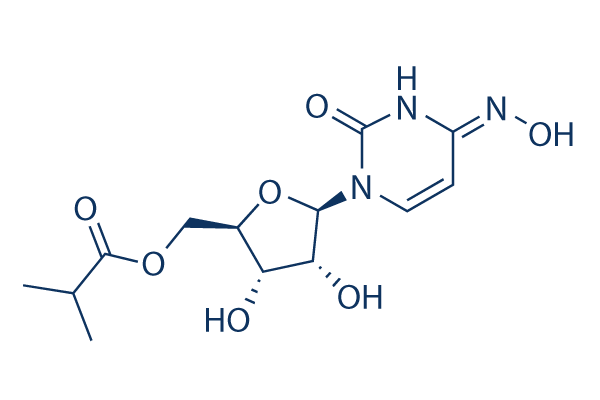
Molnupiravir Eidd 2801 99 Hplc Selleck Sars Cov Inhibitor

Antivirale Therapie Positives Und Negatives Zu Molnupiravir Bei Covid 19 Pz Pharmazeutische Zeitung

Molecular Mechanisms Of Corona Drug Candidate Molnupiravir Unraveled Max Planck Gesellschaft

A Daily Pill To Treat Covid Could Be Just Months Away Scientists Say
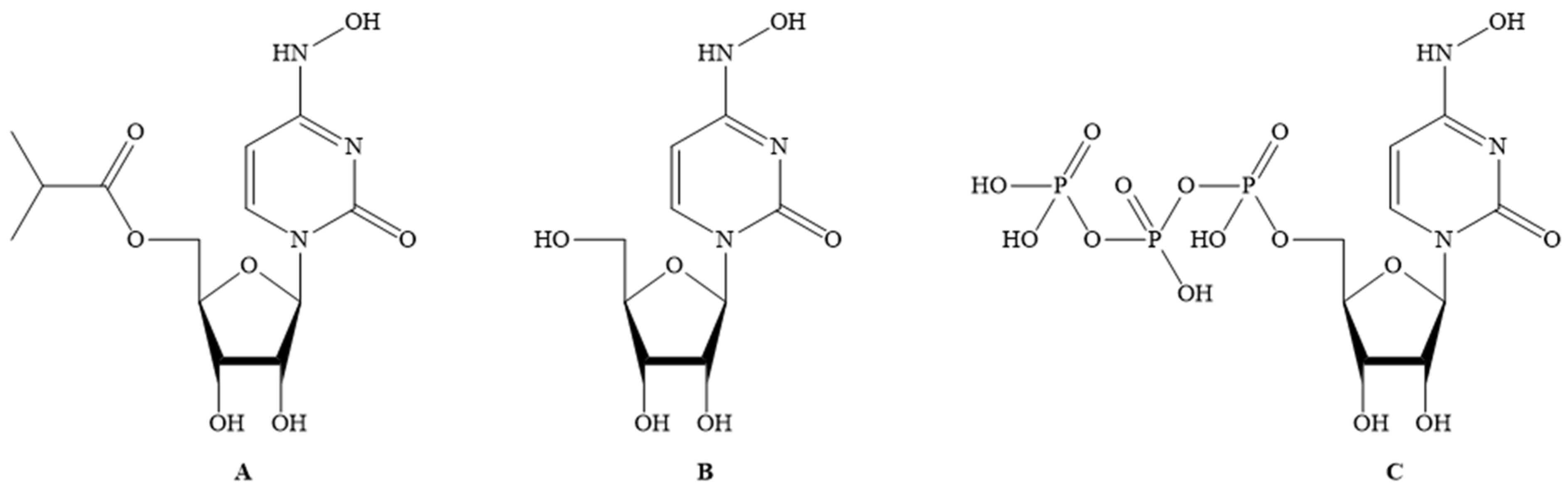
Molecules Free Full Text Discovery Development And Patent Trends On Molnupiravir A Prospective Oral Treatment For Covid 19












Post a Comment
Post a Comment